The Role of Iron in Plants
Iron (Fe) is an essential micronutrient for the growth and development of plants. The availability of iron affects the yield and quality of agricultural crops, and consequently, human nutrition.
Fe is poorly available for plant use because it is mostly present in soils in the form of insoluble oxides/hydroxides, especially at neutral or alkaline pH. Scientists have long studied how plants cope with low iron content and their ability to absorb it from the soil. Research has highlighted that plants have developed two different strategies for acquiring iron from the soil. The so-called Strategy I (iron reduction strategy) and Strategy II (iron chelation strategy).
Strategy I is used by non-graminaceous (non-grass) species, while graminaceous plants use Strategy II.

Figure 1. Strategies used by plants to absorb iron from the soil. Top — Strategy I – Reduction Strategy (characteristic of non-grass plants like tomatoes): Phenolic compounds (PC) and protons (H+) are released from the roots via transporters AHA2 and PDR9, respectively (1A, red arrows), and in the rhizosphere, they increase the availability of Fe³⁺ (2A). Fe³⁺ is converted to Fe²⁺ by the plasma membrane protein FRO2 (3A, yellow dashed arrow). Fe²⁺ enters the roots through the transporter IRT1 (4A, green arrow). Bottom — Strategy II – Chelation Strategy (used by grass plants like wheat): Phytosiderophores (PS) are released from the roots via the transporter TOM1 (1B, red arrow); in the rhizosphere, they form a complex with Fe³⁺ (2B). The PS-Fe³⁺ complex enters the roots through transporters YS1 or YSL (3B, green arrow).
It has recently been discovered that these two strategies are not entirely exclusive and that the mechanism plants use to absorb Fe is directly shaped by the characteristics of the soil they grow in (e.g., pH, oxygen concentration, etc.).
Iron (Fe) is an essential micronutrient for almost all living organisms. It is found in cofactors that ensure the activity and/or stability of metalloproteins involved in many physiological processes (e.g., respiration, photosynthesis, sulfur and nitrogen assimilation, amino acid biosynthesis).
For most organisms, a disruption in Fe homeostasis leads to cellular defects. These defects negatively affect growth.
Stress from Fe deficiency inhibits plant growth. The content of vitamin C, soluble protein, and soluble sugar in leaves and stems significantly decreases under iron deficiency conditions. Meanwhile, cellulose and nitrate levels increase.
Iron deficiency stress clearly reduces the net photosynthetic rate and nitrate reductase activity in leaves.
As a result of Fe deficiency, the balance system of active oxygen metabolism is disrupted. This leads to a decrease in catalase activity, superoxide dismutase activity in roots and leaves, and peroxidase (POD) activity in leaves. At the same time, POD activity in roots and malondialdehyde content in roots and leaves significantly increase.
In humans, disruptions in iron homeostasis are associated with several health problems, such as cancer risk, neurodegenerative diseases, and iron deficiency anemia. Fe-deficiency anemia is the most common, affecting about one billion people worldwide. Increasing the iron content in their diet would help overcome the associated symptoms. Thus, increasing the iron content and its bioavailability in crops through biofortification would have a huge beneficial impact on human health. A key step in achieving this goal is deciphering the molecular mechanisms that regulate iron homeostasis in plants.
Some studies have shown that in plants, an excess of Fe leads to plant growth defects and reduced yields. This is because, under aerobic conditions, Fe can react with hydrogen peroxide (H₂O₂) to form reactive oxygen species (ROS) through the so-called Fenton reaction, which is harmful to cells.
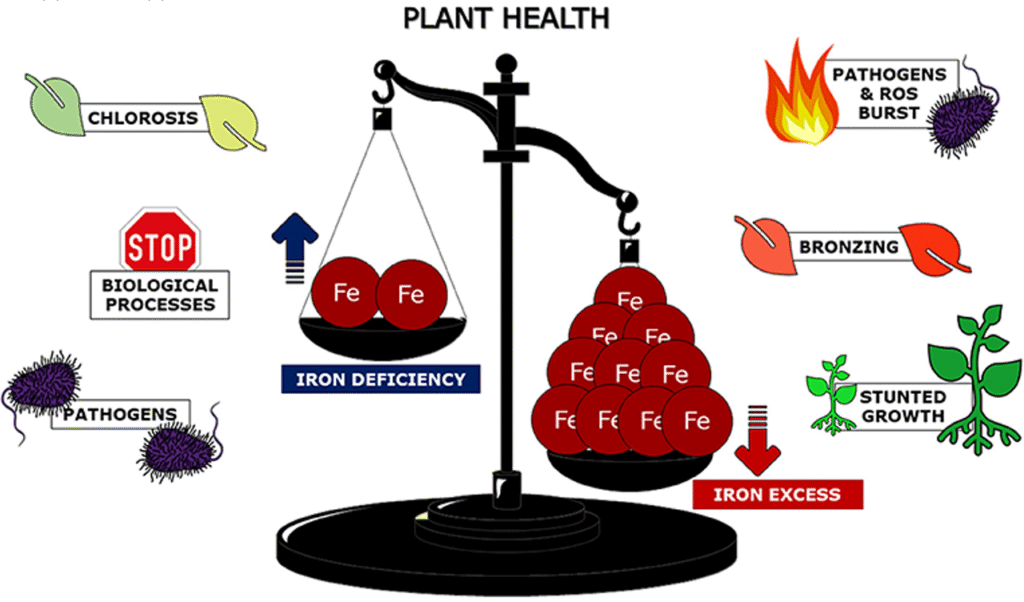
Nevertheless, iron deficiency is widespread, even though iron is the fourth most abundant element on Earth. This is because Fe is mostly present in soils in the form of poorly soluble oxides/hydroxides. Especially at neutral or alkaline pH. Therefore, iron homeostasis in plant cells must be tightly regulated to avoid its deficiency or excess. Both of which strongly affect crop yields and the quality of products derived from them.
Since iron in soils is often in forms that are not readily available to plants, applying this element through foliar feeding acts as a factor for the biofortification of plant products. For instance, research conducted in Pakistan in 2018 found that foliar application of citric acid, FeSO₄ + citric acid, and a combination of FeSO₄, citric acid, and a surfactant led to increased photosynthetic activity.
According to another study conducted in Indonesia in 2019, the application of FeSO₄ affected the number and weight of fruits.
A double application of 1% FeSO₄ at 15 and 30 days after transplanting tomato seedlings resulted in the highest number and weight of fruits. This effect was observed up to the third harvest. Iron application increased the dry matter yield of shoots and roots. It also improved plant height of sorghum, chlorophyll content in leaves, and the concentration and uptake of Fe by shoots compared to the control treatment.
Foliar feeding with Fe reduced the concentrations of P and Mn in shoots. However, it had no significant effect on the concentrations of P, Mn, and Fe in roots. These were the results of a study conducted in Iran in 2014. Given the positive effects mentioned above, foliar application of iron can benefit both the vitality of plants and the enrichment of the human diet.
We at Wonder strongly recommend adding WL Mono Fe 10 to the nutrition systems of all agricultural crops. This measure will serve as a factor in increasing the yield and quality of the final produce.
📞 Contact our agronomists for personal advice: +38 067 0000 304
📩 Or write to us at: info@wonder-corporation.com — we are available and ready to help!




















